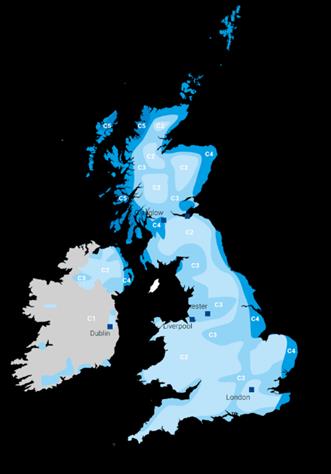
This causes metals closer towards the ocean to rust and corrode faster than those more inland.
Water in our oceans is recorded at an average 3.5% salinity. This means that salts, such as sodium chloride (NaCl), comprise roughly 35 parts per thousand (ppt) of seawater. While this may sound like a low-level density, chlorides exhibit an extremely aggressive affinity to metals and can rapidly penetrate steel grades that are not suitably corrosion resistant. Chlorides eat into the material causing spots and pits in the component surface, which can propagate through the material and rapidly cause complete mechanical failure. This phenomenon is known as pitting corrosion.
Metals like stainless steel and titanium are often exposed to seawater corrosion. Rust can be prevented with a process called galvanisation. This process adds a protective layer of zinc to the surface of metal.